Ageing is universal. Every human being experiences it, and every biological system changes over time. But in medicine and public discussion there is persistent confusion about where normal ageing ends and disease begins. The distinction matters. It influences clinical decision making, public health policy, research priorities, and how individuals interpret changes in their own bodies.
Understanding the boundary between ageing and disease requires examining how biological systems change across the lifespan, why some changes are considered normal adaptations while others are classified as pathology, and how modern science is redefining these categories.
What Is Biological Ageing?
Biological ageing refers to the gradual decline in physiological function that occurs with increasing chronological age. These changes affect nearly every organ system and arise from cumulative molecular and cellular processes.
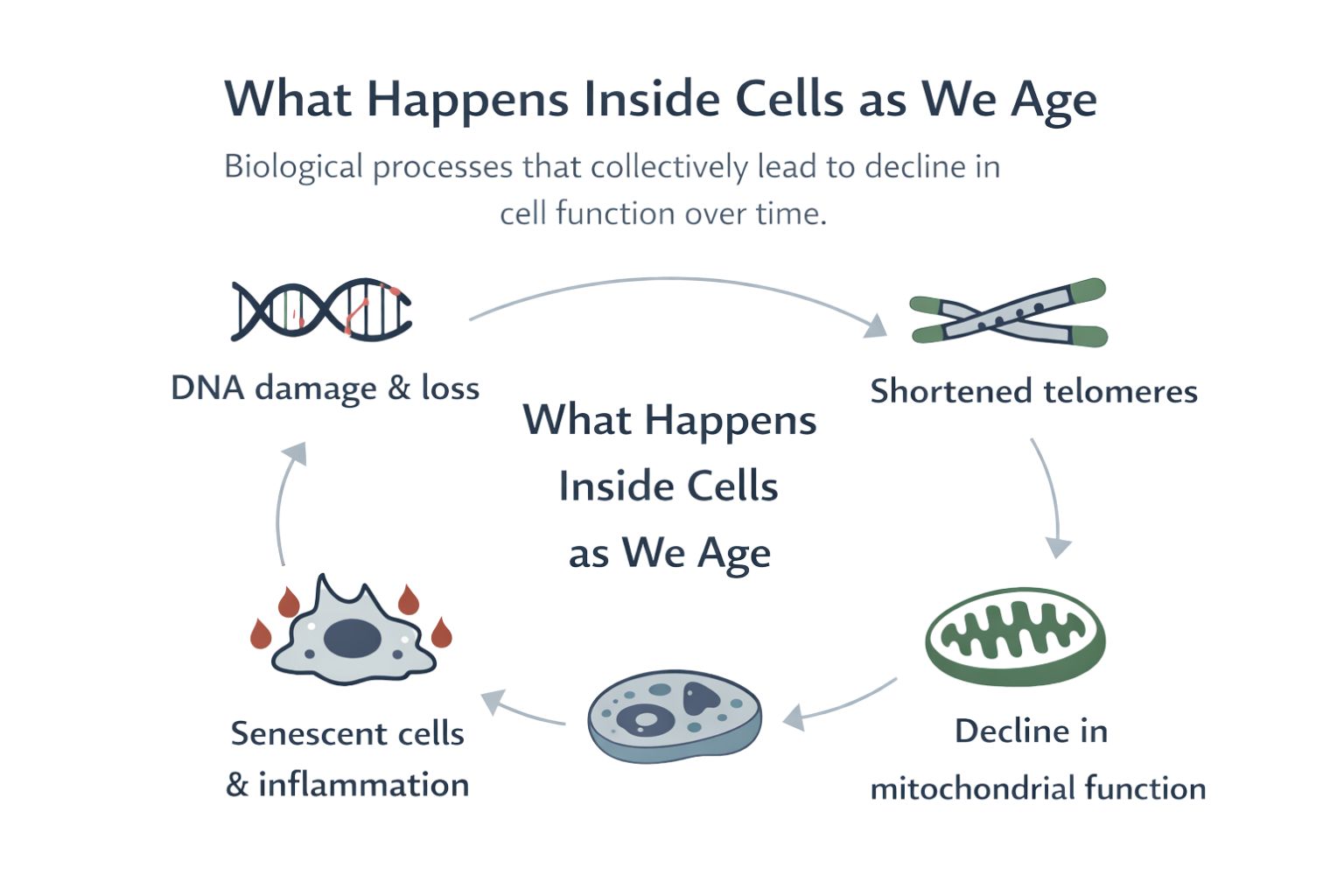
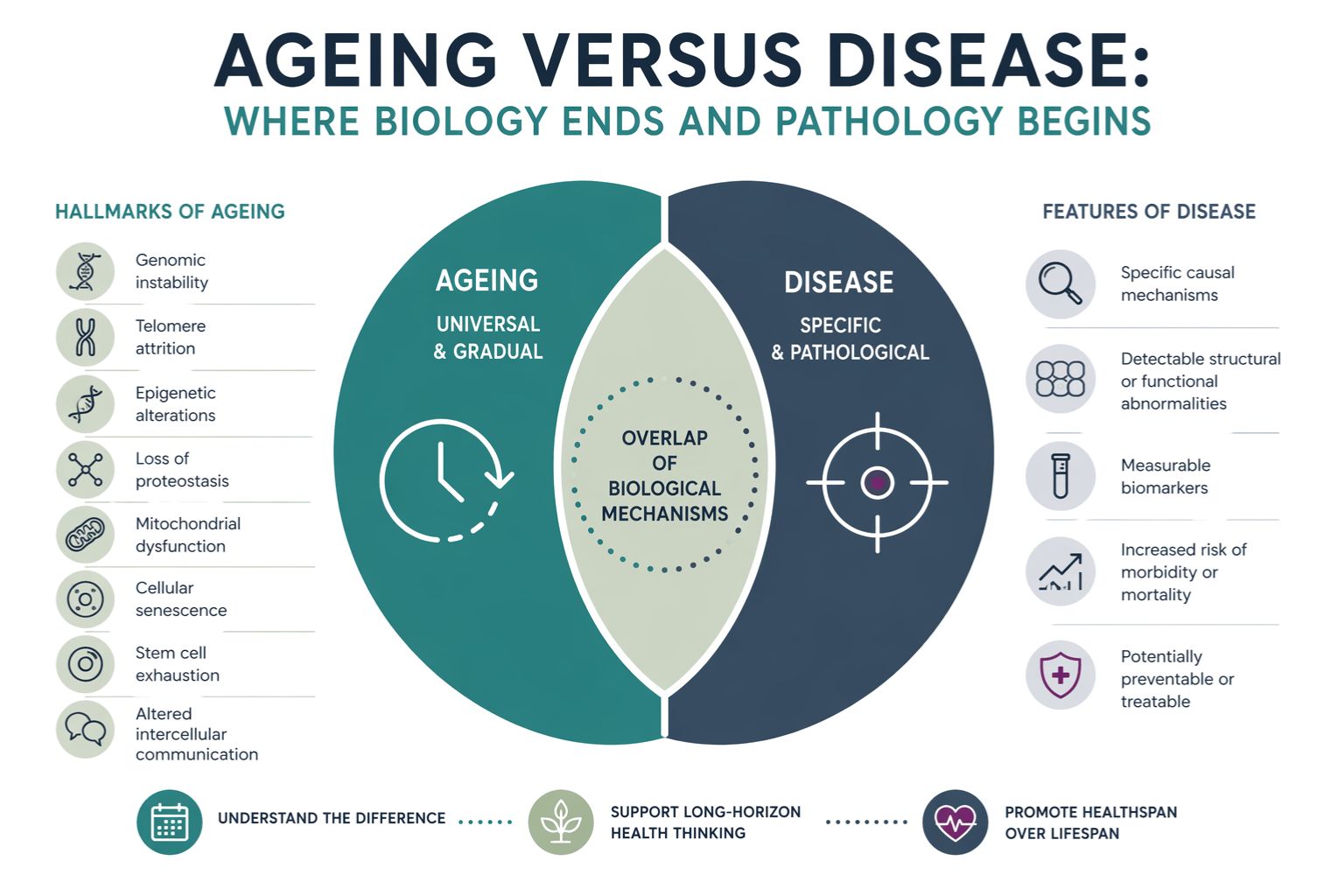
At the cellular level, ageing involves several interconnected mechanisms often described as the hallmarks of ageing. These include genomic instability, telomere shortening, epigenetic alterations, mitochondrial dysfunction, cellular senescence, impaired protein homeostasis, altered nutrient sensing, stem cell exhaustion, and changes in intercellular communication.
These processes do not occur suddenly. They accumulate slowly across decades and influence how tissues maintain themselves, repair damage, and respond to environmental stress.
For example, the immune system gradually shifts with age. T cells become less diverse, inflammatory signalling increases, and the capacity to respond to novel pathogens declines. This phenomenon, often described as immunosenescence, illustrates how ageing modifies physiology without necessarily constituting disease.
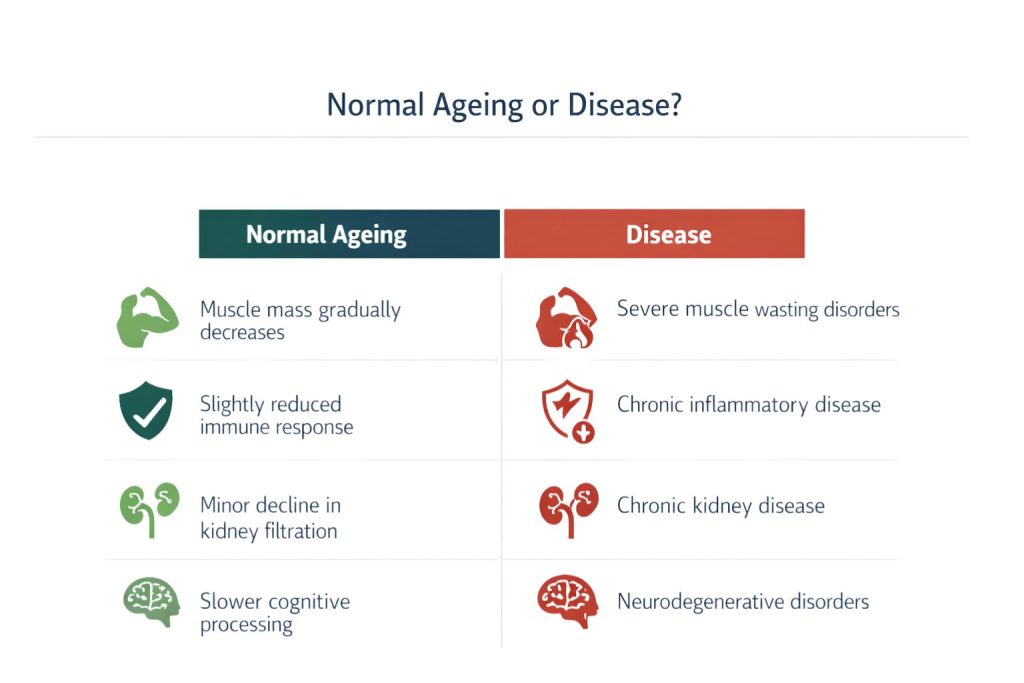
Similarly, muscle mass decreases slowly with age, a process known as sarcopenia. Bone density gradually falls. Skin becomes thinner and less elastic. These are expected biological changes that occur in most individuals as part of normal ageing.
However, not all age related changes should be interpreted as inevitable or benign. Some reflect underlying disease processes that become more common with age.

Defining Disease
In medicine, disease is typically defined as a condition in which normal physiological processes are disrupted in ways that impair health or function. Diseases usually involve identifiable pathological mechanisms such as infection, inflammation, genetic mutation, metabolic dysregulation, or structural damage to tissues.
Examples include cancer, diabetes, Alzheimer disease, and cardiovascular disease. These conditions have specific causes, diagnostic criteria, and treatment strategies.
The challenge arises because many diseases become more common with age. Age is the single strongest risk factor for many major illnesses. As a result, the biological processes associated with ageing often overlap with mechanisms that contribute to disease.
This overlap raises a critical question. When biological deterioration occurs gradually over time, how should scientists and clinicians determine whether the change represents normal ageing or the early stages of pathology?
The Continuum Between Ageing and Disease
Rather than being completely separate categories, ageing and disease exist on a continuum. Many biological pathways that contribute to ageing also participate in the development of disease.
For example, chronic low level inflammation increases with age. This phenomenon is sometimes referred to as inflammageing. Elevated inflammatory signalling contributes to the development of multiple conditions including atherosclerosis, neurodegeneration, and metabolic disease.
Similarly, mitochondrial dysfunction is a hallmark of ageing. As mitochondria become less efficient at producing energy, cells accumulate oxidative damage. This process contributes both to normal ageing and to diseases such as Parkinson disease and cardiovascular disease.
Another example involves cellular senescence. Senescent cells stop dividing but remain metabolically active, releasing inflammatory molecules that affect surrounding tissues. Small numbers of these cells may play useful roles in wound healing and tumour suppression. However, when senescent cells accumulate excessively they can promote tissue degeneration and chronic disease.
These examples demonstrate that the biological mechanisms underlying ageing can also drive pathology. The difference between ageing and disease often lies in degree, timing, and context rather than completely distinct processes.
Physiological Decline Versus Pathological Damage
One way to distinguish ageing from disease is to consider whether the biological change represents a gradual decline in function or specific structural or molecular damage.
Normal ageing often involves reduced physiological reserve. Organs remain functional but have less capacity to respond to stress. For instance, the heart of an older adult may pump effectively under normal conditions but struggle to adapt to intense physical demand.
Disease, by contrast, typically involves identifiable damage or dysfunction. In coronary artery disease, fatty plaques accumulate in blood vessels and restrict blood flow. In osteoarthritis, cartilage within joints degenerates and structural changes develop in bone.
However, the distinction is not always straightforward. Mild reductions in kidney filtration rate are common in older adults and may represent normal ageing. But a more severe decline can signal chronic kidney disease. Determining where the boundary lies requires careful clinical evaluation and often depends on the degree of impairment.
Biological Variability in Ageing
Another important factor is that ageing does not occur at the same rate in every individual. Two people of the same chronological age may have very different biological profiles.
Some individuals remain physically and cognitively robust well into their eighties or nineties. Others develop chronic diseases decades earlier. Genetics, environment, lifestyle, and social factors all influence how ageing unfolds.
This variability complicates attempts to define clear biological boundaries between ageing and disease. A physiological change that appears normal in one individual may represent early pathology in another.
Modern research increasingly focuses on biological age, which attempts to measure the functional state of tissues rather than simply counting years since birth. Biomarkers such as DNA methylation patterns, inflammatory markers, and metabolic indicators are being investigated as potential measures of biological ageing.
These tools may eventually help clinicians distinguish between healthy ageing and early disease processes more precisely.
Ageing as a Risk Landscape
From a scientific perspective, ageing can be understood as a landscape of increasing vulnerability. As biological systems gradually lose resilience, the likelihood of disease rises.
For example, ageing affects the integrity of blood vessels. Over time, endothelial cells become less responsive to regulatory signals, arterial walls stiffen, and lipid metabolism changes. These shifts create conditions in which atherosclerosis can develop more easily.
Similarly, age-related changes in DNA repair mechanisms allow genetic mutations to accumulate in cells. While many mutations remain harmless, some can initiate the development of cancer.
In this sense, ageing does not directly cause disease but creates an environment in which disease becomes more probable.
When Ageing Itself Becomes Pathological
There are situations in which the ageing process itself appears to drive disease more directly. Neurodegenerative disorders provide a clear example.
Conditions such as Alzheimer disease involve the accumulation of abnormal proteins in the brain, including beta amyloid and tau. While these proteins are associated with pathological damage, their accumulation is also linked to age-related changes in cellular maintenance systems.
Proteostasis mechanisms that normally remove misfolded proteins become less efficient with age. As a result, harmful protein aggregates can build up in neurons. The boundary between normal age-related decline in cellular maintenance and pathological accumulation of proteins becomes blurred.
Researchers are increasingly exploring whether targeting the fundamental biology of ageing could delay or prevent multiple diseases simultaneously. If successful, such approaches could shift the focus of medicine from treating individual diseases to maintaining biological resilience.
Medical Classification and Ethical Considerations
Another dimension of the ageing versus disease debate concerns how societies choose to classify age-related changes.
If ageing itself were formally defined as a disease, it could open the door to new regulatory pathways for therapies aimed at slowing ageing. Pharmaceutical companies might develop drugs targeting ageing mechanisms, and clinical trials could measure biological ageing markers as outcomes.
However, many scientists and ethicists argue that labelling ageing as a disease could have unintended consequences. Ageing is a natural biological process experienced by everyone. Defining it as pathological might medicalise normal human experience.
Instead, some researchers advocate focusing on healthspan, the period of life during which individuals remain healthy and functional. From this perspective, the goal is not to eliminate ageing but to reduce the burden of disease and disability associated with it.
Clinical Implications
For clinicians, distinguishing ageing from disease is essential for appropriate medical care. Not every change observed in older adults requires intervention.
For example, slight reductions in hearing or vision may reflect normal sensory ageing. Aggressive treatment may not be necessary unless these changes significantly impair daily life.
Conversely, attributing symptoms to ageing when they actually reflect disease can delay diagnosis and treatment. Fatigue, weight loss, memory impairment, or persistent pain should not automatically be dismissed as normal consequences of growing older.
Effective healthcare requires careful assessment of biological function, medical history, and individual risk factors. Understanding the underlying mechanisms of ageing helps clinicians recognise when physiological decline crosses the threshold into pathology.
The Future of Ageing Research
Advances in molecular biology, genetics, and computational analysis are transforming the study of ageing. Researchers are now able to examine cellular processes in unprecedented detail and track how they change over time.
New experimental approaches are investigating interventions that target ageing mechanisms directly. These include drugs that influence nutrient sensing pathways, compounds that remove senescent cells, and therapies that enhance mitochondrial function.
While much of this research remains in early stages, it reflects a growing recognition that the biology of ageing plays a central role in many major diseases.
Understanding where ageing ends and disease begins is therefore not merely an academic question. It shapes the future of medicine.
Conclusion
Ageing and disease are deeply interconnected but not identical. Ageing involves gradual biological changes that reduce physiological resilience. Disease involves specific pathological processes that disrupt normal function.
The boundary between the two is not fixed. Instead, it reflects a complex interaction of molecular mechanisms, environmental influences, and individual variation.
Modern science increasingly recognises that many diseases arise from biological systems that have lost the capacity to maintain stability over time. By studying the mechanisms of ageing, researchers hope to identify strategies that preserve health and function throughout the lifespan.
Clarifying the biological boundaries between ageing and disease remains one of the most important challenges in biomedical science. The answer will influence how medicine approaches prevention, treatment, and the pursuit of healthier lives across an ageing global population.