Ageing Versus Disease: Where Biology Ends and Pathology Begins
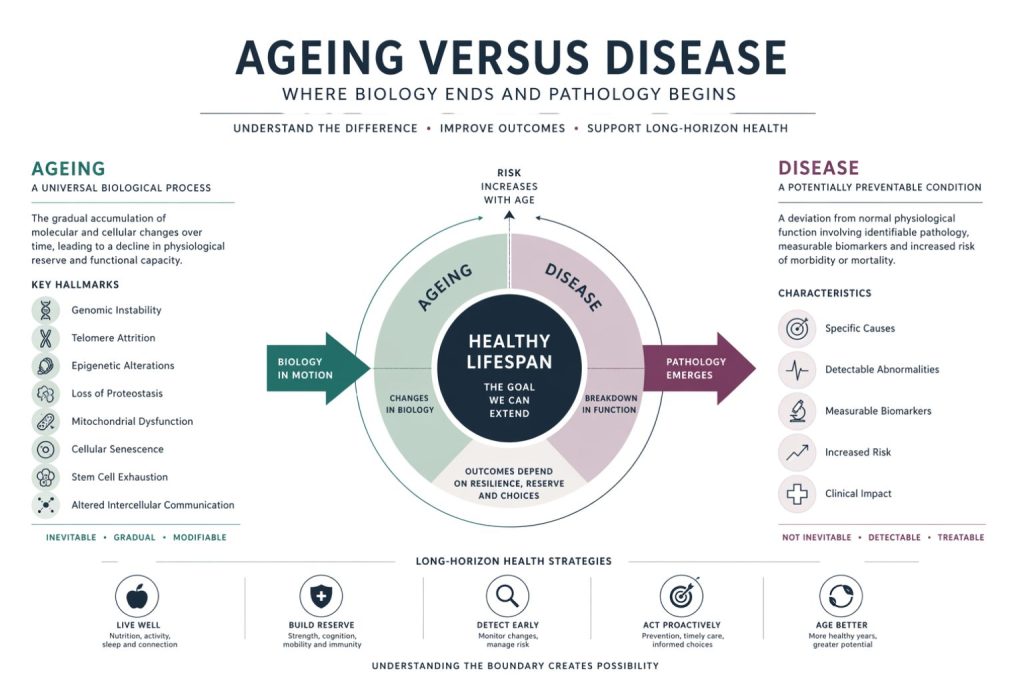
Ageing is universal. Disease is not. Yet, in clinical practice and public discourse, the two are frequently conflated. Wrinkles, reduced mobility, memory lapses, and declining physiological reserves are often treated as inevitable consequences of growing older, rather than as potentially modifiable processes or early indicators of pathology. This lack of distinction has significant implications. It shapes how healthcare systems allocate resources, how research priorities are set, and how individuals interpret changes in their own bodies.
A scientifically rigorous understanding of where normal biological ageing ends and disease begins is therefore essential. It enables more precise diagnostics, more effective prevention strategies, and a reframing of ageing itself not as decline, but as a dynamic biological process that can be influenced.
This article examines that boundary. It explores the mechanisms of ageing, the defining characteristics of disease, and the grey zone where the two intersect.
Defining Biological Ageing
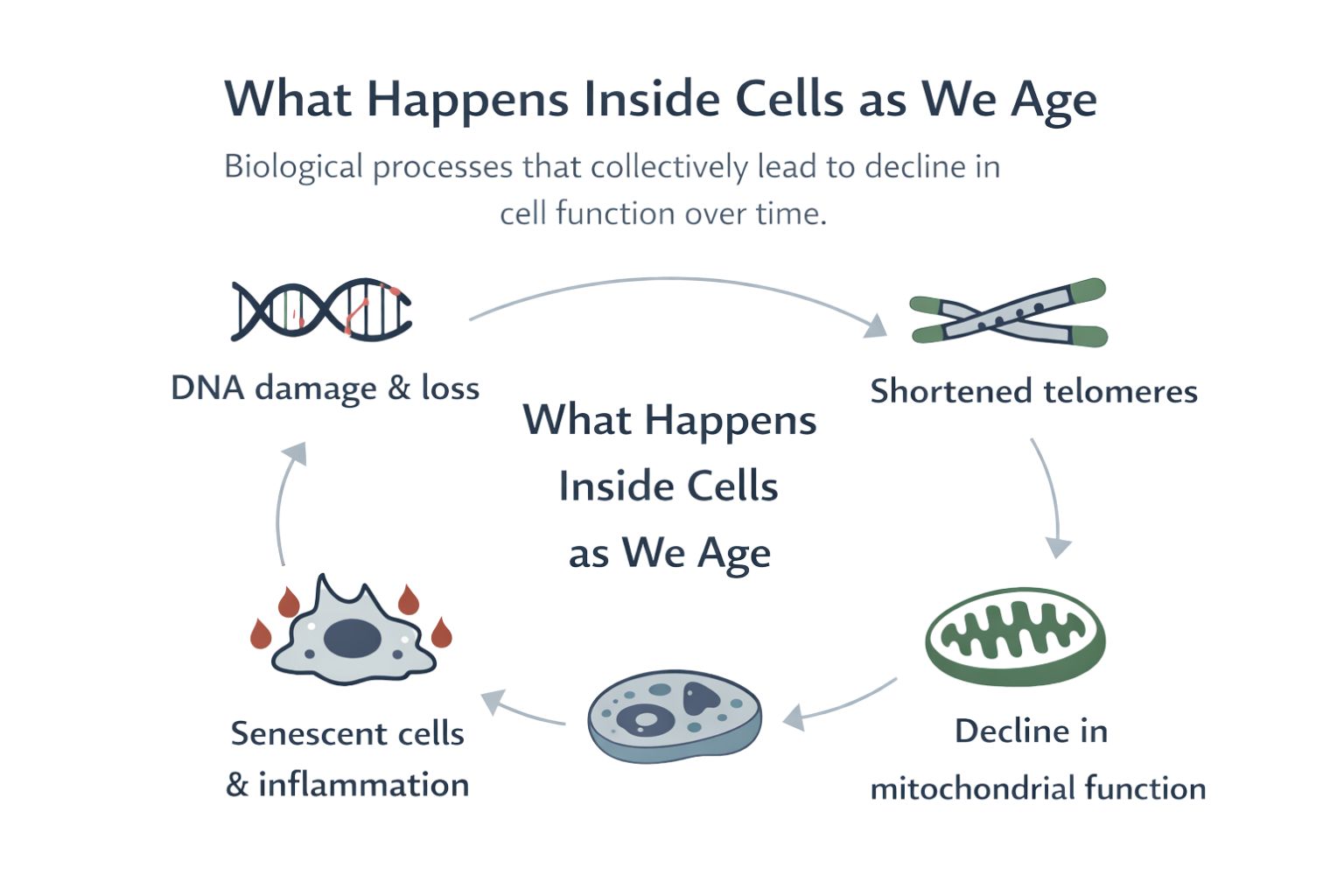
Biological ageing refers to the progressive accumulation of molecular and cellular damage over time, leading to a gradual decline in physiological function. It is not a single process, but a network of interrelated mechanisms.
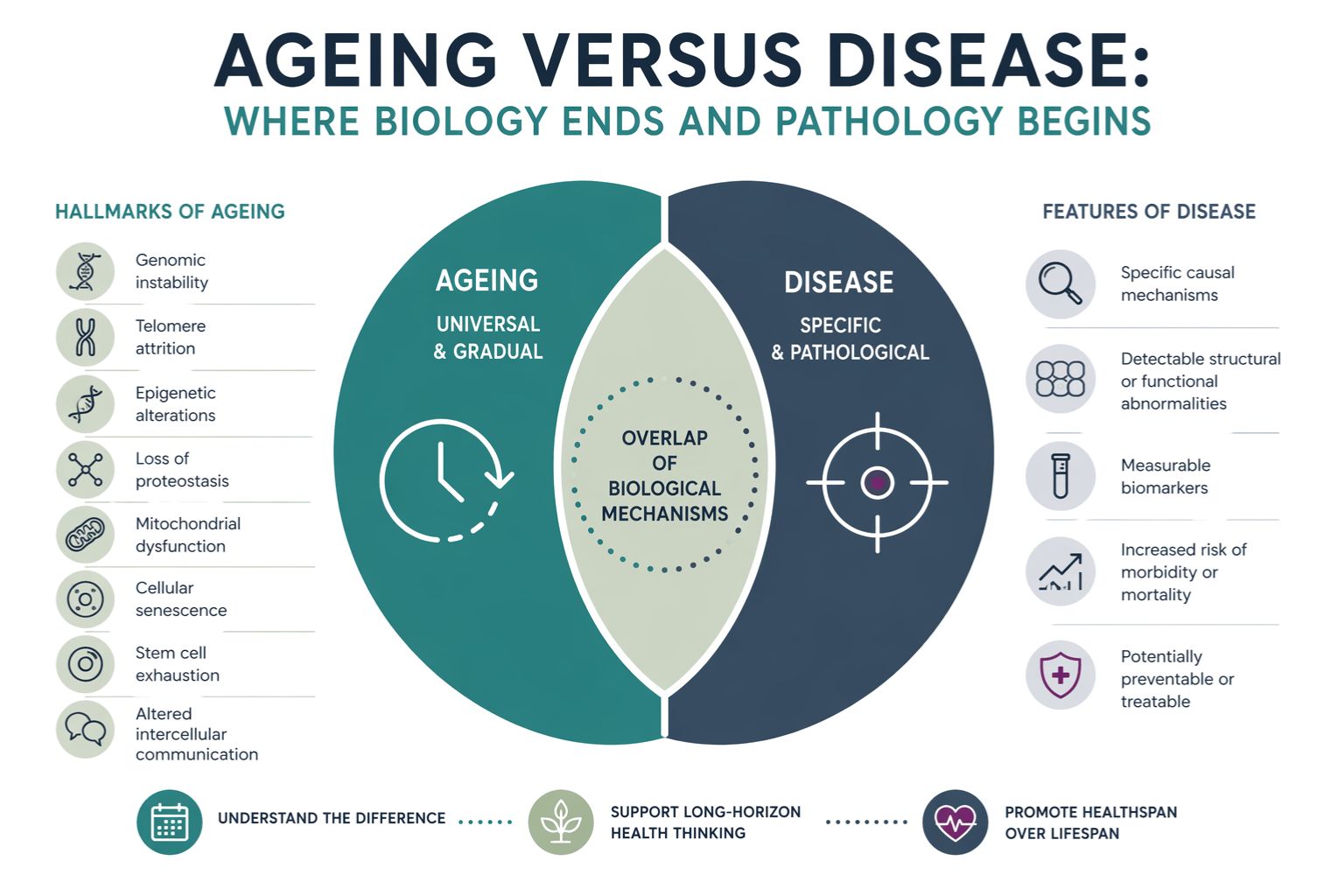
Key hallmarks of ageing include:
- Genomic instability
- Telomere attrition
- Epigenetic alterations
- Loss of proteostasis
- Mitochondrial dysfunction
- Cellular senescence
- Stem cell exhaustion
- Altered intercellular communication
These processes occur in all individuals, regardless of lifestyle or environment, although the rate at which they progress varies significantly. Importantly, biological ageing is not synonymous with chronological age. Two individuals of the same age may exhibit markedly different biological profiles.
Ageing, in this context, is a baseline process. It is universal, gradual, and intrinsic to living systems.
Defining Disease
Disease, by contrast, is characterised by a deviation from normal physiological function that results in identifiable pathology and, typically, clinical symptoms. It is often associated with:
- Specific causal mechanisms
- Detectable structural or functional abnormalities
- Measurable biomarkers
- Increased risk of morbidity or mortality
Diseases can be acute or chronic, infectious or non-infectious, genetic or environmental in origin. Crucially, disease is not inevitable. While risk increases with age, disease represents a breakdown or dysregulation beyond what is expected from normal ageing alone.
This distinction is central. Ageing increases vulnerability. Disease reflects failure of resilience.
The Overlap Between Ageing and Disease
The challenge lies in the fact that many diseases associated with later life emerge from the same biological processes that drive ageing.
For example:
- Mitochondrial dysfunction contributes to both normal ageing and neurodegenerative conditions
- Chronic low-grade inflammation, often termed “inflammageing,” is a feature of ageing but also underpins cardiovascular disease and type 2 diabetes
- Cellular senescence plays a role in tissue repair but, when accumulated, contributes to cancer and fibrosis
This overlap creates a continuum rather than a clear boundary. Ageing provides the biological context in which disease develops.
However, overlap does not imply equivalence. The presence of a shared mechanism does not mean that the outcome is inevitable or indistinguishable.
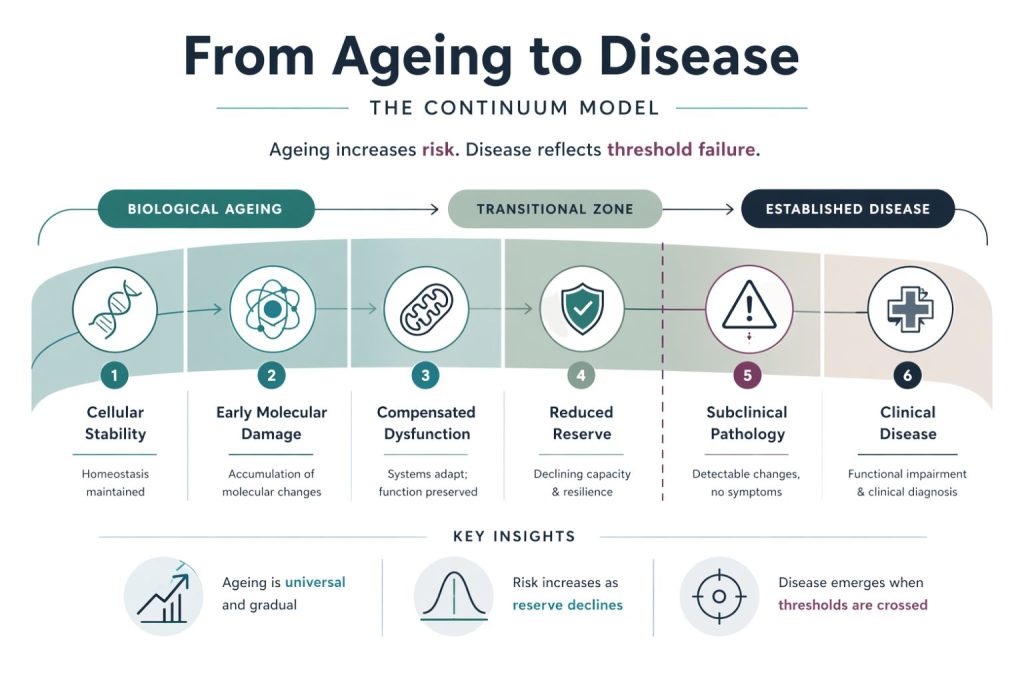
Physiological Decline Versus Pathological Change
A useful framework for distinguishing ageing from disease is to consider the difference between physiological decline and pathological change.
Physiological Decline
This includes gradual, predictable changes such as:
- Reduced maximal cardiac output
- Decreased bone density
- Slower cognitive processing speed
- Decline in muscle mass and strength
These changes occur across populations and do not necessarily impair function to a clinically significant degree. They represent a shift in baseline capacity rather than a failure of the system.
Pathological Change
By contrast, pathological changes involve:
- Structural damage, such as atherosclerotic plaques
- Cellular degeneration beyond expected levels
- Dysregulated signalling pathways
- Loss of functional independence
For example, mild memory lapses may be consistent with ageing, whereas progressive cognitive decline affecting daily functioning is indicative of a neurodegenerative condition.
The distinction hinges on severity, progression, and impact on function.
Biomarkers and the Limits of Measurement
Modern medicine increasingly relies on biomarkers to distinguish normal ageing from disease. These include:
- Blood-based markers such as inflammatory cytokines
- Imaging findings
- Genetic and epigenetic profiles
- Functional assessments
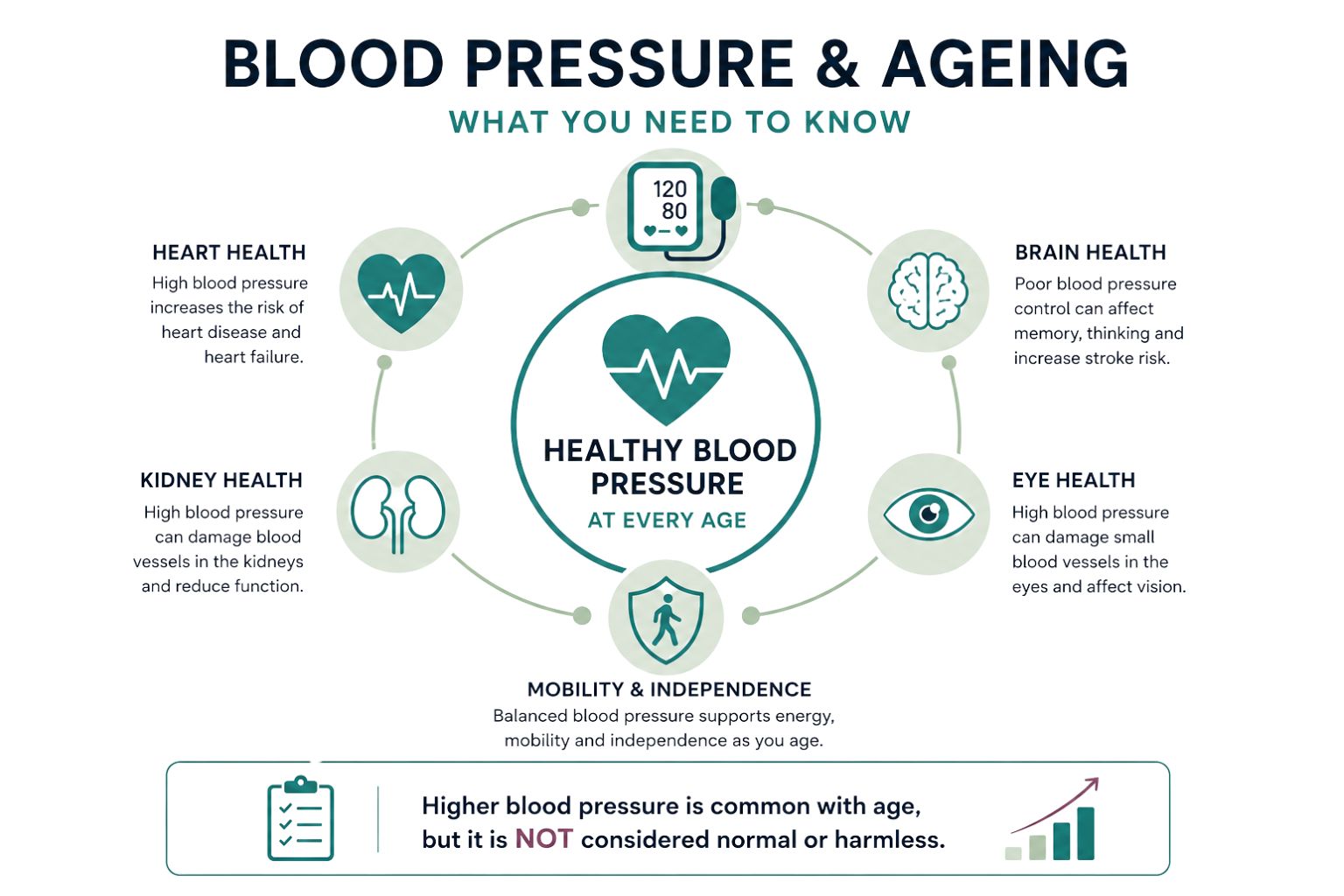
However, biomarkers are not definitive. Many exist on a spectrum, and thresholds for “normal” versus “abnormal” are often based on population averages rather than individual trajectories.
For instance, elevated blood pressure may be common in older populations, but it is not a benign feature of ageing. It reflects vascular changes that increase the risk of cardiovascular events.
Similarly, the presence of amyloid plaques in the brain may precede symptoms of dementia by years or even decades. Whether this represents early disease or an aspect of ageing remains a subject of ongoing research.
The limitation is clear. Measurement does not always resolve the conceptual boundary.
The Role of Resilience and Reserve
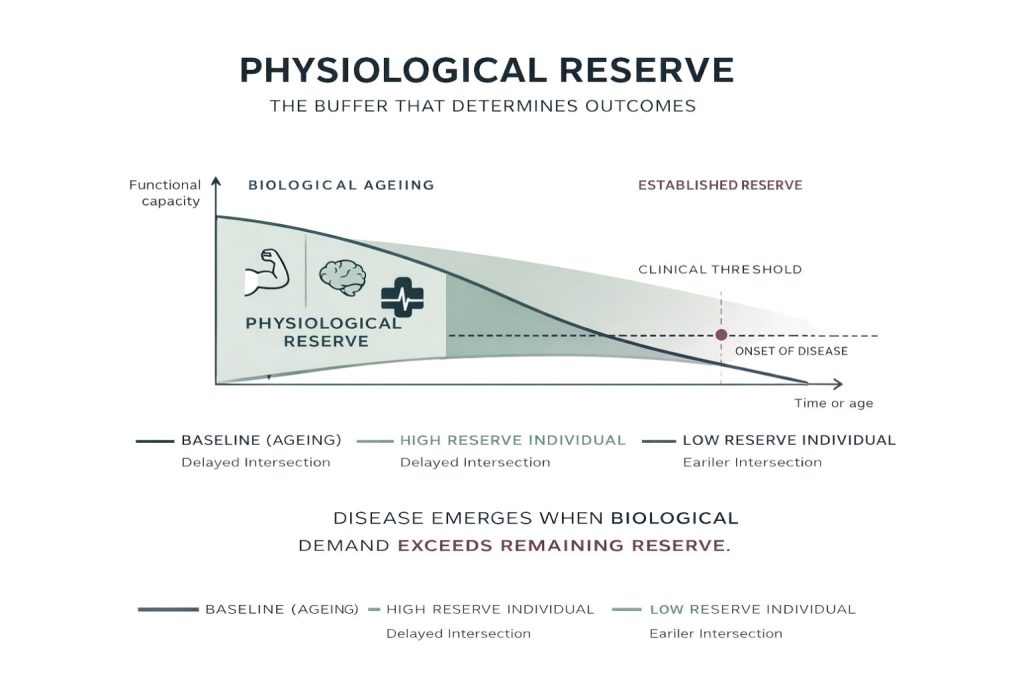
One of the most important distinctions between ageing and disease lies in the concept of physiological reserve.
Reserve refers to the capacity of an organ or system to withstand stress and maintain function. With age, reserve declines. However, individuals with higher reserve can tolerate greater levels of damage without developing clinical disease.
This is evident in multiple systems:
- Cardiovascular reserve determines how well the heart responds to increased demand
- Cognitive reserve influences the ability to compensate for neuronal loss
- Immune reserve affects the response to infection
Disease often emerges when reserve is exceeded. Ageing reduces the buffer. Disease occurs when the threshold is crossed.
This perspective shifts the focus from static definitions to dynamic capacity.
Risk, Probability, and Determinism
A critical misconception is the assumption that age-related risk equates to inevitability. While the probability of many diseases increases with age, this does not mean that disease is an intrinsic component of ageing.
For example:
- Not all older adults develop cardiovascular disease
- Not all experience significant cognitive decline
- Not all develop cancer
This variability reflects the interaction between genetic predisposition, environmental exposure, lifestyle factors, and stochastic biological events.
From a scientific standpoint, ageing is associated with increased risk, not deterministic outcomes. Disease remains contingent.
Implications for Healthcare
The distinction between ageing and disease has practical consequences for healthcare systems.
Prevention Versus Acceptance
If certain conditions are incorrectly classified as “normal ageing,” opportunities for prevention may be missed. For example:
- Treating hypertension as inevitable delays intervention
- Accepting frailty as unavoidable limits rehabilitation efforts
- Normalising cognitive decline reduces early detection of dementia
A more precise understanding supports proactive care.
Resource Allocation
Healthcare systems must decide where to allocate resources. Blurring the line between ageing and disease can lead to either over-medicalisation or under-treatment.
- Over-medicalisation risks unnecessary interventions
- Under-treatment risks neglect of modifiable conditions
Balance requires clarity.
Research Priorities
Ageing research increasingly focuses on targeting the underlying biological processes rather than individual diseases. This includes:
- Senolytic therapies aimed at clearing senescent cells
- Interventions targeting metabolic pathways
- Strategies to reduce chronic inflammation
These approaches recognise that modifying ageing biology may delay or prevent multiple diseases simultaneously.
Ethical Considerations
There is also an ethical dimension. Defining ageing as disease could lead to:
- Expansion of medical intervention into normal life processes
- Increased healthcare costs
- Shifts in societal expectations around longevity
Conversely, failing to recognise treatable conditions as disease may result in inequitable care.
The boundary is not purely scientific. It reflects values, priorities, and definitions of health.
A Systems Perspective
From a systems biology perspective, ageing and disease are best understood as interconnected processes within a complex adaptive system.
Ageing alters the system’s baseline state:
- Increased noise in biological signalling
- Reduced efficiency of repair mechanisms
- Greater susceptibility to perturbation
Disease represents a state in which the system moves beyond adaptive capacity into dysfunction.
This framework avoids binary classification. Instead, it emphasises trajectories, thresholds, and system dynamics.
Supporting Long Horizon Health Thinking
For individuals and policymakers, the distinction between ageing and disease informs long horizon health strategies.
Focus on Trajectory, Not Snapshot
Health should be viewed as a trajectory over time rather than a single measurement. Early deviations from optimal function may signal increased risk long before disease manifests.
Invest in Modifiable Factors
While ageing itself cannot be stopped, many contributing factors can be influenced:
- Nutrition
- Physical activity
- Sleep
- Social engagement
- Environmental exposures
These factors affect both the rate of ageing and the likelihood of disease.
Maintain Reserve
Strategies that preserve physiological and cognitive reserve are central:
- Strength training to maintain muscle mass
- Cognitive stimulation to support brain function
- Preventive healthcare to manage emerging risks
The aim is not to eliminate ageing, but to extend the period of functional health.
Conclusion
The boundary between ageing and disease is neither fixed nor simple. It is a continuum shaped by biological processes, individual variability, and contextual factors.
Ageing is a universal, intrinsic process characterised by gradual decline in physiological function. Disease represents a deviation from this baseline, marked by dysfunction, pathology, and increased risk of adverse outcomes.
The overlap between the two reflects shared mechanisms, but it does not erase the distinction. Understanding where biology ends and pathology begins requires attention to function, progression, and resilience.
This distinction matters. It influences how health is defined, how care is delivered, and how individuals approach their own ageing.
A scientifically grounded perspective enables a shift from passive acceptance to informed intervention. It supports a model of ageing that is not synonymous with decline, but one that recognises the potential to shape outcomes across the lifespan.